-
FDA Panel Says Bisphosphonates Need Label Changes on Use Duration

This is the recommendation that came down today although the Food and Drug Administration does not have to accept the recommendations of its panels.Bone drugs from Warner Chilcott Plc, Roche Holding AG, Merck & Co. and Novartis AG need labeling changes to reduce the risk of fractures, a U.S. panel said.
The companies should add clarifications on the length of time that osteoporosis patients may take the medicines, outside advisers to the Food and Drug Administration said today in a 17- 6 vote in Adelphi, Maryland. The FDA isn’t required to follow its panels’ recommendations.
The agency has evaluated the safety of the drugs, known as bisphosphonates, for almost four years and cited possible links to unusual thigh fractures and jawbone deterioration in 2010. The agency said in July it also was examining conflicting studies on whether bisphosphonate pills such as Warner Chilcott’s Actonel, Merck’s Fosamax and Roche’s Boniva raise esophageal cancer risks.
A revised label should “be very clear that efficacy may fall off after a period of time, perhaps five years,” panelist Lewis Nelson, director of the medical toxicology fellowship program at New York University, said after the vote. “Serious concerns have been raised about risk, and those need to be continually evaluated as well.”
There needs to be additional research, period.
Mere warning labels are not going to answer the questions from every day patients – how long do I take the medicine and what protocol do I use? Or, what is the chance my femur will fracture or will I develop osteonecrosis of the jaw (ONJ) if I have a tooth removed.
-
Oral Hygiene and Antibiotics Play a Role in Healing and Pain Alleviation in Bisphosphonate-Related Osteonecrosis of the Jaw (BRONJ)
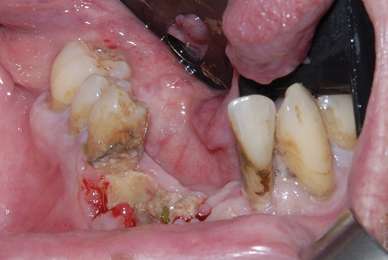
Osteonecrosis of the jaw in a patient who had poor oral hygiene and generalized periodontal disease and recently underwent routine dental extractions in the mandible. This patient had undergone monthly intravenous bisphosphonate therapy for treatment of multiple myeloma during the previous 12 months.
Photo credit: Mayo ClinicAccording to this new paper here.
Abstract
OBJECTIVES:
The objectives of this study were to define the incidence, pain, and healing in cancer patients treated with intravenous bisphosphonates.
STUDY DESIGN:
The study included long-term follow-up of 99 bisphosphonate-using patients (group A) and conservative treatment of 67 patients with bisphosphonate-related osteonecrosis of the jaw (BRONJ, group B) using 3 antibiotic schemes and oral hygiene.
RESULTS:
The frequency of zoledronic acid single-agent use was 85.9% and 69.8% in group A and B, respectively. Median follow-up was 13 months (group A) and 16 months (group B). Two patients in group A developed BRONJ (2%). Of those with BRONJ in group B who completed follow-up, healing occurred in 14.9% (7/47) and pain subsided in 80.9% (38/47). Healing was significant in patients who received pamidronate followed by zoledronic acid (P = .023) and with BRONJ stages 0 and stage I (P = .003).
CONCLUSIONS:
This case series suggests that oral hygiene and conservative antibiotic therapy play a role in healing and pain alleviation in BRONJ. Oral hygiene and follow-up may decrease incidence of BRONJ.
This sounds a reasonable course, knowing that this complication will be found to occur with increasing frequency. Let us hope a conservative protocol can be developed for these unfortunate patients.
Previous:
Jury Finds Novartis the Manufacturers of Zometa Not Liable for Osteonecrosis of the JawOral Bisphosphonates: Study – Absolute Risk for Femur Fracture Low with Bisphosphonates
Revisiting Bisphosphonates and Femur Fractures
New Dentistry Cause for Alarm for Patients Who Use Bisphosphonates – Fosamax, Actonel, Boniva?
-
Jury Finds Novartis the Manufacturers of Zometa Not Liable for Osteonecrosis of the Jaw

Osteonecrosis of the Jaw (ONJ)
Another bisphosphonate law suit.A U.S. federal jury found that a bone drug made by Novartis Pharmaceuticals Corp was not to blame for the severe jaw deterioration developed by a Rhode Island man who died of cancer in 2005.
Karleen Hogan, the widow of Timothy Hogan, claimed in a lawsuit filed in 2006 that Novartis had failed to properly warn her husband about the severe adverse effects caused by Zometa, a drug used to strengthen bones in cancer patients. She sought compensatory damages for her husband’s suffering.
The suit is one of an estimated 600 filed against the unit of Novartis AG in recent years blaming the company for suppressing information about adverse effects linked to Zometa and Aredia, another bone-strengthening drug.
I think what helped the defendants in this case was the testimony from the patient’s physican and dentist who said he had pre-exisiting dental problems and that the benefits of the drug outweighed the risks.
But, there is a mixed record on these suits and there will be more to come.
Hogan’s case was initially consolidated with hundreds of Zometa and Aredia liability suits in multi-district litigation in Tennessee federal court. Similar litigation is also pending in a state court in New Jersey.
Hogan’s is the fourth Zometa case to go to trial. In October 2010, a New Jersey superior court jury ruled in favor of Novartis. A jury in Montana state court awarded a plaintiff with the same jaw disease $3.2 million in October 2009, and in November, a federal jury in North Carolina awarded a North Carolina woman’s family $12.8 million, later reduced to $1.26 million.
Osteonecrosis of the Jaw Associated With Bisphosphonate Agent Zoledronic Acid and Chemotherapy Combined With the Antiangiogenic Agent Bevacizumab
Oral Bisphosphonates: Study – Absolute Risk for Femur Fracture Low with Bisphosphonates
Revisiting Bisphosphonates and Femur Fractures
New Dentistry Cause for Alarm for Patients Who Use Bisphosphonates – Fosamax, Actonel, Boniva?
-
Osteonecrosis of the Jaw Associated With Bisphosphonate Agent Zoledronic Acid and Chemotherapy Combined With the Antiangiogenic Agent Bevacizumab

Osteonecrosis of the Jaw (ONJ)
This study has just appeared in the Journal of the American Dental Association. Here is the abstract.Background. The authors investigated the incidence of and risk factors for osteonecrosis of the jaw (ONJ) in patients with metastases to the bone who received the bisphosphonate agent zoledronic acid (ZOL) and chemotherapy combined with the antiangiogenic agent bevacizumab (BEV).
Methods. The authors evaluated 59 participants (34 with breast cancer and 25 with nonsmall-cell lung cancer). All of the participants received 4 milligrams of ZOL via intravenous (IV) infusion every four weeks and 15 mg per kilogram of BEV every three weeks. They conducted a dental examination in participants at baseline and every three months until the patients died or were lost to follow-up. If needed, participants received periodontal disease treatment and underwent tooth extraction before they started receiving ZOL and BEV.
Results. The median time the participants received ZOL therapy was 18.8 months (range, 3.1–28.9 months); 36 participants (61.0 percent) received ZOL therapy for more than one year. The median time participants received BEV therapy was 16.7 months (range, 2.8–29.6 months). None of the participants required dentoalveolar surgery while undergoing cancer treatment. After a median follow-up period of 19.7 months, none of the participants developed bisphosphonate-related ONJ.
Conclusions and Clinical Implications. ZOL combined with BEV did not predispose to ONJ participants with cancer that had metastasized to the bone who underwent a baseline dental examination and preventive dental measures. The study results must be considered in the context of the study’s protocols and the follow-up period.So, the question is whether these patients were prevented from developing osteonecrosis of the jaw (ONJ) because of the drug therapy or because of dental treatment protocols?
The researchers took several measures to reduce the study participants’ risk of developing ONJ, including the following:
- Dental caries and periodontal disease were treated before starting study treatment.
- Mouth rinses with chlorhexidine and local antibiotic agents were administered before baseline oral hygiene.
- Recommendations were made for maintaining good oral hygiene.
- Teeth were extracted at least four weeks before starting ZOL and BEV therapy.
- Invasive dental procedures were avoided during treatment.
- If invasive dental procedures were needed during treatment, ZOL and BEV were readministered after at least four weeks.
All the patients received a dental exam and panoramic x-rays before starting treatment and every three months until the patients died or were lost to follow-up. After a median follow-up period of 19 months, none of the study participants had developed ONJ.
So, antiangiogenesis or treating the patient dentally prior to IV ZOL treatment?
The jury continues to be out.
But, preventive dental measures, including baseline dental examinations and follow-up are simple to institute and probably prudent, in any case.
“Although further research is needed, the results of our study suggest that ZOL combined with the antiangiogenic agent BEV does not predispose patients with metastases to the bone from breast and NSCL cancer to ONJ if they undergo a baseline dental examination,” they concluded. “Nevertheless, the results of the study must be considered in the context of the follow-up period used in the study and the use of the preventive dental protocol.”
Tanja Fehm, MD, from the department of obstetrics and gynecology at the University of Tübingen in Germany, has done similar research (Gynecologic Oncology, March 2009, Vol. 112:3, pp. 605-609). She told DrBicuspid.com that the incidence of ONJ is low — between 1% and 4% — in metastatic breast cancer patients receiving bisphosphonates.
“Therefore, the number of patients (59) investigated in this study is too low to make meaningful conclusions,” she added. “However, the paper summarizes the preventive measures that can help avoid ONJ.”
Previous:
Oral Bisphosphonates: Study – Absolute Risk for Femur Fracture Low with Bisphosphonates
Revisiting Bisphosphonates and Femur Fractures
New Dentistry Cause for Alarm for Patients Who Use Bisphosphonates – Fosamax, Actonel, Boniva?