-
Fosamax Osteonecrosis of the Jaw Lawsuit Set for Trial September 7

Osteonecrosis of the Jaw (ONJ)
Another oral bisphosphonate (Fosamax with ONJ) lawsuit trial set.The next trial date for a Fosamax lawsuit involving jaw necrosis is scheduled to begin early next month.
The Fosamax trial will involve a complaint brought by Linda Secrest, of Florida, who alleges that Merck failed to adequately warn that side effects of Fosamax, the popular osteoporosis drug, can lead to severe jaw bone decay. Trial is scheduled to begin on September 7, in the U.S. District Court for the Southern District of New York.
Fosamax is an osteoporosis drug that belongs to a family of similar medications known as bisphosphonates. Long-term use of oral bisphosphonates has been linked to an increased risk of serious and debilitating jaw problems, known as osteonecrosis of the jaw. The condition causes the jaw bone to decay and rot, often resulting in the need for surgery to remove portions of the jaw.
Merck currently faces more than 1,100 Fosamax jaw lawsuits, most of which have been consolidated and centralized for pretrial proceedings in the U.S. District Court for the Southern District of New York as part of an MDL or multidistrict litigation.
There have already been several trials for Fosamax jaw necrosis lawsuits that have been held in the federal MDL over the last two years. While Merck has successfully defended its medication in three cases, one lawsuit resulted in an $8 million jury award for Fosamax jaw damage last year, finding that Merck failed to adequately research the potential side effects of warn about the risk of jaw necrosis from Fosamax.
A few more cases and Merck will probably propose some industry-wide settlement agreement.
Next up will be the lawsuits over bone fractures from Fosamax.
-
Oral Hygiene and Antibiotics Play a Role in Healing and Pain Alleviation in Bisphosphonate-Related Osteonecrosis of the Jaw (BRONJ)
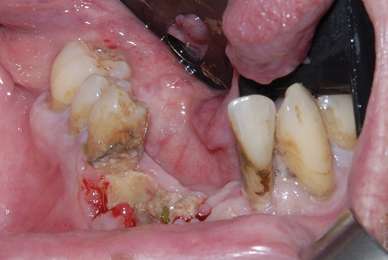
Osteonecrosis of the jaw in a patient who had poor oral hygiene and generalized periodontal disease and recently underwent routine dental extractions in the mandible. This patient had undergone monthly intravenous bisphosphonate therapy for treatment of multiple myeloma during the previous 12 months.
Photo credit: Mayo ClinicAccording to this new paper here.
Abstract
OBJECTIVES:
The objectives of this study were to define the incidence, pain, and healing in cancer patients treated with intravenous bisphosphonates.
STUDY DESIGN:
The study included long-term follow-up of 99 bisphosphonate-using patients (group A) and conservative treatment of 67 patients with bisphosphonate-related osteonecrosis of the jaw (BRONJ, group B) using 3 antibiotic schemes and oral hygiene.
RESULTS:
The frequency of zoledronic acid single-agent use was 85.9% and 69.8% in group A and B, respectively. Median follow-up was 13 months (group A) and 16 months (group B). Two patients in group A developed BRONJ (2%). Of those with BRONJ in group B who completed follow-up, healing occurred in 14.9% (7/47) and pain subsided in 80.9% (38/47). Healing was significant in patients who received pamidronate followed by zoledronic acid (P = .023) and with BRONJ stages 0 and stage I (P = .003).
CONCLUSIONS:
This case series suggests that oral hygiene and conservative antibiotic therapy play a role in healing and pain alleviation in BRONJ. Oral hygiene and follow-up may decrease incidence of BRONJ.
This sounds a reasonable course, knowing that this complication will be found to occur with increasing frequency. Let us hope a conservative protocol can be developed for these unfortunate patients.
Previous:
Jury Finds Novartis the Manufacturers of Zometa Not Liable for Osteonecrosis of the JawOral Bisphosphonates: Study – Absolute Risk for Femur Fracture Low with Bisphosphonates
Revisiting Bisphosphonates and Femur Fractures
New Dentistry Cause for Alarm for Patients Who Use Bisphosphonates – Fosamax, Actonel, Boniva?
-
New Dentistry Cause for Alarm for Patients Who Use Bisphosphonates – Fosamax, Actonel, Boniva?
Cross Posted from Flap’s Dentistry BlogA new study coming out of Flap’s alma mater, the University of Southern California School of Dentistry has reported an increased incidence in the occurrence of osteonecrosis of the jaw among patients taking oral bisphosphonates, including Fosamax, Actonel and Boniva.In a separate report Thursday in the Journal of the American Dental Assn., Parish Sedghizadeh of the USC School of Dentistry reported that he had observed nine cases of osteonecrosis of the jaw among 208 patients taking oral bisphosphonates — an unusually high incidence.
Osteonecrosis of the jaw — death of the bone — is known to occur in 1% to 10% of cancer patients taking intravenous bisphosphonates to combat bone weakening brought on by chemotherapy. It is rarely observed in patients taking oral forms of the drug, however.
Some people immediately dismissed his findings, arguing that the widespread use of the drugs would have already revealed such a high incidence of a disabling side effect.
“The most important criticism is that the sample size was only 208 patients,” said Dr. Robert R. Recker, director of the Osteoporosis Research Center at Creighton University in Omaha and president of the National Osteoporosis Foundation. “The study design and sample size were simply inadequate to make any conclusions.”
Both the American Society for Bone and Mineral Research and the American Dental Assn. have recently conducted comprehensive reviews of the risks of osteonecrosis associated with oral bisphosphonates, added Dennis Black, a professor of epidemiology and biostatistics at UC San Francisco. They concluded “that the risk is somewhere between 1 in 10,000 and 1 in 100,000, and it is not clear that this is increased by bisphosphonates,” he said.
The American Dental Association today issued the following advisory which is here.
Most noteworthy from the advisory:
- The American Dental Association has reviewed previous studies on this issue which indicate that use of oral bisphosphonates (medication commonly prescribed for patients with osteoporosis) has been associated with a low risk for developing ONJ-one study indicated 1 out of 2,260 people taking oral bisphosphonates develop ONJ. This new study from USC claims that the frequency of ONJ among oral bisphosphate users is higher than previously reported-approximately 4 percent of the people studied.
- It’s important for me to know if you are on bisphosphonate drugs or if you’ve used them in the past because of the possible risk of developing ONJ. ONJ is uncommon, but it can be serious. If you have received bisphosphonate therapy intravenously related to cancer therapy, you may be at a higher risk of developing ONJ than if you take oral medication.
- Current ADA recommendations suggest that routine dental treatment generally need not be altered merely because a patient is taking or has taken oral bisphosphonates. However, patients with any history of bisphosphonate therapy should be especially encouraged to practice optimal oral hygiene and to receive routine dental examinations. Furthermore, a comprehensive oral examination and treatment before or soon after commencing bisphosphonates therapy may be beneficial for patients if they’re not already receiving regular dental care to treat existing oral health problems
A previously issued patient handout is available for download here (pdf).
This is a controversy which will be further studied.
In the meantime, Flap urges caution for patients taking ORAL Bisphosphonate medications. And,please patients update your health history and tell your dentist if you are using these drugs.
Previous:
Technorati Tags: Bisphosphonate, Fosamax, Actonel, Boniva, Osteonecrosis of the Jaw